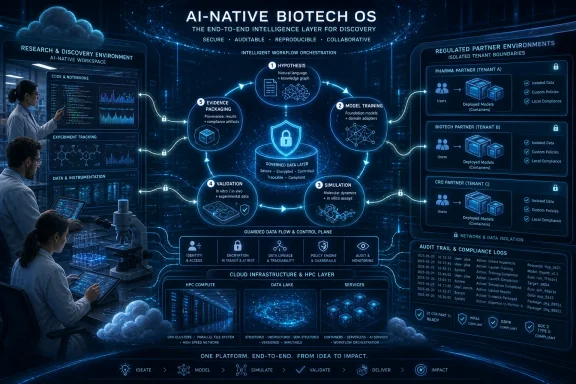
Microsoft is pitching Azure and Microsoft for Startups as the foundation for an AI-native biotech operating model in 2026, arguing that young life-sciences companies need cloud-scale compute, secure model deployment, reproducible workflows, and agentic research tooling before they can credibly partner with pharma or regulators. The argument is not subtle: biotech’s next platform war will be fought less over who has the best model demo and more over who can turn messy science into governed, repeatable evidence. That is a more interesting claim than a startup marketing post usually makes, because it captures a real shift now underway across computational biology, drug discovery, and clinical-data work. Microsoft wants founders to see “platform” not as infrastructure plumbing, but as the operating system of the company itself.
The old startup-cloud pitch was simple: take credits, spin up infrastructure, and worry about enterprise architecture later. That bargain worked tolerably well for web apps, SaaS dashboards, and consumer experiments, where the first job was to prove product-market fit before the bills got serious. Biotech has never quite fit that model, because the “product” is often evidence, and evidence becomes less valuable when its lineage is murky.
Microsoft’s new biotech framing recognizes that difference. The company is saying, in effect, that the startup stack now has to satisfy three audiences at once: scientists who need speed, engineers who need operational sanity, and external partners who need proof that sensitive data and regulated processes are under control. That is not merely a better Azure brochure. It is a bet that infrastructure choices will increasingly determine whether a biotech startup looks like a credible partner or an expensive science project.
There is a reason the language sounds like an operating-system metaphor. In modern biotech, the work is not just a sequence of experiments; it is a loop of hypothesis generation, model training, simulation, validation, documentation, and collaboration. If that loop lives across notebooks, ad hoc scripts, unmanaged clusters, and one-off file transfers, the company accumulates technical debt at the exact moment it is trying to accumulate scientific credibility.
Microsoft’s pitch is therefore aimed at a pain point founders already understand. The problem is not that biotech startups lack ambition. It is that a small team can be asked to behave like a pharmaceutical-grade computational organization before it has the headcount, procurement muscle, or process maturity to do so.
That is why hyperscaler strategy matters in life sciences in a way it does not always matter elsewhere. A biotech startup running large simulations, training specialized models, analyzing clinical data, or collaborating with pharma is not merely renting servers. It is choosing the substrate on which its claims about reproducibility, security, and scalability will later be judged.
The Microsoft blog’s strongest point is that scientific ambition increasingly collides with infrastructure reality. Frontier workloads are bursty. A team may spend days iterating interactively, then suddenly need massive compute for simulation, molecular screening, model training, or evaluation runs. Hiring a full internal platform team too early can kill runway; postponing platform discipline too long can make the science hard to reproduce.
This is the paradox Microsoft is trying to exploit. Startups want the freedom of improvisation, but their customers and partners want the reassurance of discipline. The cloud provider that can make serious workflows feel less like bespoke infrastructure engineering has a stronger claim on the future biotech customer than one that merely offers the longest catalog of services.
Microsoft’s article leans into this reality by emphasizing iteration, fine-tuning, evaluation, lineage, and reproducibility. In biotech, generic models may help with literature review, coding, summarization, or workflow automation, but competitive advantage usually depends on proprietary data and specialized modeling choices. That means the startup’s system must support custom architectures, secure training and post-training, and controlled deployment paths rather than treating AI as a commodity API call.
The phrase AI-native gets abused in startup marketing, but here it has a concrete meaning. An AI-native biotech company is one whose experimental and computational loops are designed around models from the beginning. Its knowledge base, validation strategy, compute patterns, and governance processes are part of the same machinery.
That machinery also has to admit uncertainty. Model evaluation in science is not a leaderboard game. A metric can look impressive while masking dataset leakage, weak external validity, poor calibration, or a mismatch between academic benchmarks and biological usefulness. When investor pressure and publication incentives enter the room, the temptation to overstate model performance becomes structural.
This is where platform architecture becomes a form of scientific hygiene. A system that records how data moved, which model version produced which output, what evaluation protocol was used, and which assumptions were baked into a run is doing more than satisfying auditors. It is protecting the company from fooling itself.
That changes the architecture. A startup may need to deploy a containerized model inside a customer’s cloud tenant, keep the customer’s data in place, protect its own intellectual property, and still scale compute reliably. That is a harder problem than serving an API from a central account. It forces the startup to think about tenant boundaries, identity, secrets, logging, update mechanisms, and operational support long before a typical early-stage company wants to.
Microsoft’s advantage, if it can execute, is that Azure already lives inside many enterprise and regulated environments. That does not automatically make Azure the right answer for every biotech workload, but it does make the argument legible to buyers who care about data residency, identity integration, compliance posture, and procurement familiarity. The platform decision becomes a sales-enablement decision as much as a technical one.
The same logic applies to clinical and patient-derived data. Once protected health information enters the workflow, the cheerful language of AI experimentation gives way to access controls, audit trails, data minimization, and governance. Agentic systems raise the stakes further, because a tool that can plan, execute, and chain actions across data sources is also a tool that can create new failure modes if permissions and boundaries are sloppy.
This is the part of the AI boom that startup pitch decks often skate past. Autonomy is not free. The more agency a system has, the more important it becomes to define what it can touch, what it can change, what it can export, and who is accountable when it produces a plausible but wrong result.
That distinction matters. A chat interface can help a scientist move faster, but it does not by itself create an R&D operating model. Discovery is designed around agents that can reason over knowledge, plan work, use tools, run analyses, and feed results back into an iterative process. The ambition is to move from “AI helps me write code” to “AI helps coordinate the research process.”
The opportunity is obvious. Scientific organizations are drowning in fragmented knowledge: papers, experimental records, internal reports, simulation outputs, assay results, datasets, and tacit process knowledge spread across teams. A graph-based knowledge engine, if implemented well, could give agents richer context than a pile of documents in a vector store. It could also make the reasoning trail more inspectable, which matters in environments where “the model said so” is not an acceptable explanation.
But the risk is equally obvious. The more Microsoft Discovery looks like an operating layer for R&D, the more customers will ask whether it can be trusted with the messy, consequential, regulated parts of science. Preview products can inspire imagination; production scientific systems have to survive procurement, validation, incident response, and skeptical domain experts.
For biotech startups, the practical question is not whether Microsoft Discovery is magical. It is whether the platform points toward a stack they should prepare for: agents as workflow participants, HPC as an on-demand substrate, proprietary data as a governed knowledge layer, and evaluation as a continuous process rather than a pre-demo ritual.
GxP is shorthand for “good practice” quality guidelines and regulations across regulated life-sciences environments. For a biotech startup, the key point is not that a cloud provider can make the company compliant by association. It cannot. The point is that a platform with independently reviewed processes, documentation, controls, and regulated-workload posture can reduce friction when a startup must answer hard questions from pharma, CROs, clinical partners, investors, or quality teams.
That reduction in friction can be strategically meaningful. Early-stage companies do not have infinite cycles to spend on security questionnaires, infrastructure explanations, audit evidence, and bespoke deployment discussions. Every week spent reconstructing how a result was generated is a week not spent improving the underlying science.
This is why trust can become a speed advantage. A startup that can show clear lineage, controlled access, reproducible workflows, and a plausible regulated-workload path may move faster through partner diligence than one with a more impressive model but a weaker operational story. The market does not always reward the cleanest science first; it often rewards the company that can make its claims usable by institutions.
Microsoft’s framing is self-serving, of course. Azure is the product being sold. But the underlying claim is right: in biotech, the operational wrapper around AI increasingly determines whether AI can leave the demo environment and enter the enterprise.
A five-person computational team cannot afford to spend months reinventing secure infrastructure patterns. A founder cannot personally referee every data-access decision once collaborations multiply. A machine-learning scientist should not have to become a part-time cloud networking specialist simply to run distributed workloads at the frontier.
This is where the operating-model language earns its keep. The question is not whether Azure has a service for every problem. The question is whether a startup can assemble a coherent way of working from those services without drowning in configuration, integration, and governance overhead. Platforms win when they compress complexity without hiding the controls that regulated customers need.
There is a delicate balance here. Too little abstraction leaves scientists stuck in infrastructure glue. Too much abstraction creates black boxes that serious partners will not trust. The winning biotech platform has to make the common path easy while preserving enough transparency for audit, validation, and expert challenge.
Microsoft’s message to founders is that these choices should be made early. That is sound advice, but it cuts both ways. Choosing a hyperscaler deeply is a form of dependency, and dependency has costs. The same architecture that makes a startup easier to partner with in one enterprise ecosystem may make it harder to stay portable later.
This is not unique to biotech. Every industry is watching AI push cloud providers from infrastructure vendors into process vendors. The difference is that science exposes the stakes more clearly. A hallucinated slide summary is annoying; a poorly governed model-driven discovery pipeline can distort research priorities, waste lab resources, or produce claims that crumble under external scrutiny.
Microsoft is not alone in seeing this opportunity. Other hyperscalers and specialist platforms are also chasing life sciences with managed data environments, research clouds, AI tooling, and compliance-oriented services. The market will not be won by press releases about agents. It will be won by the tedious details: identity integration, container deployment, data controls, cost predictability, observability, validation support, and the ability to meet scientists where they actually work.
Still, Microsoft has a distinctive strategic opening. Its enterprise footprint gives it a familiar route into pharma and regulated organizations. Its AI partnership and Azure AI stack give it model-building credibility. Its productivity software gives it a natural surface area for knowledge work. Microsoft Discovery attempts to connect those assets into something more vertical than generic cloud infrastructure.
That is the bigger story behind the startup blog. Microsoft is not merely asking biotech founders to apply for credits. It is trying to define what a credible AI-first biotech company should look like — and then position Azure as the place where that company is easiest to build.
Microsoft’s post wisely avoids telling founders simply to “adopt agents.” The better advice is to architect for agentic workflows deliberately. That means treating transparency, traceability, permissions, containment, and human oversight as design requirements rather than compliance afterthoughts.
A useful scientific agent should be able to show its work. It should expose which data it used, which tools it called, which assumptions it made, and where human approval entered the loop. It should be constrained by role and context, not handed broad access because the demo looked impressive.
This will matter even more as agents move from code generation and workflow automation into semi-autonomous execution. In a biotech setting, an agent might generate analysis scripts, select datasets, trigger simulations, summarize literature, prepare reports, or suggest next experiments. Each of those tasks can save time. Each can also introduce errors that look polished enough to pass casual review.
The lesson for founders is straightforward: autonomy without governance is not a platform advantage. It is a liability waiting for diligence.
An evidence package includes results, but it also includes context. What data was used? What transformations were applied? What model version generated the output? What evaluation criteria were chosen? What failed? What was repeated? What can an external party inspect without compromising intellectual property?
This is where many AI-first companies outside life sciences have been allowed to be vague. Biotech does not offer the same luxury. If a startup wants to influence a development program, support a clinical path, or persuade a pharma partner to run deeper validation, it needs more than a confident dashboard. It needs a chain of custody for scientific claims.
The irony is that this kind of discipline can feel slow at the beginning. Founders want velocity, and process feels like drag. But poor provenance creates a balloon payment later, when a partner asks for evidence and the team has to reconstruct the past from notebooks, Slack threads, storage buckets, and memory.
Microsoft’s platform argument is strongest when it frames reproducibility not as bureaucracy but as acceleration. If evidence is the product, then the system that produces, records, and packages evidence is core product infrastructure.
The scientific workstation is becoming part of a larger governed fabric. Researchers may still live in Python, notebooks, specialized tools, and lab systems, but the environment around them increasingly resembles enterprise IT. That creates opportunities for sysadmins and cloud architects who understand both operational discipline and user friction.
It also creates a warning. If IT turns agentic science into a locked-down maze, researchers will route around it. If researchers ignore governance, partners and regulators will eventually force a reckoning. The winning organizations will be the ones that make the secure path the usable path.
Microsoft’s biotech message is therefore part of a broader Microsoft story. The company wants Azure, identity, security, AI tooling, and domain-specific platforms to become the default control plane for high-value work. In biotech, that work happens to be drug discovery, clinical evidence, and scientific R&D. In other sectors, the same pattern will show up as engineering, finance, legal operations, manufacturing, and design.
That does not mean startups should avoid hyperscaler-native design. In many cases, avoiding it is the riskier move. A fragile platform assembled from unmanaged scripts and heroic engineering effort is not more independent in any meaningful sense. It is merely dependent on people and practices that may not scale.
But founders should be clear-eyed. The right platform strategy preserves leverage where it matters. Containerized workloads, well-defined data boundaries, exportable evidence, documented evaluation pipelines, and clean interfaces can help a startup benefit from Azure without turning every future partnership into a rewrite.
This is especially important in pharma, where the customer’s cloud environment may not match the startup’s preferences. A credible “run where the data lives” story should not be an afterthought. It should be part of the product architecture from the beginning, because enterprise adoption often depends on deployment patterns as much as model performance.
Microsoft is correct that platform is strategy. The corollary is that platform strategy should be negotiated, not sleepwalked into.
The core takeaways are practical rather than mystical:
Biotech’s AI future will not be decided by the most theatrical agent demo or the most extravagant claim about replacing scientists. It will be decided by which companies can turn computation into credible evidence again and again, under the scrutiny of partners who own the data, regulators who demand traceability, and scientists who know how easily biology humiliates overconfident software. Microsoft’s wager is that Azure can become the operating system for that discipline; founders should treat the pitch seriously, but not passively, because the platform choices they make now may define not only how fast they move, but whether anyone believes the results when they arrive.
Source: Microsoft Biotech’s AI upgrade: An operating system built for science
 Microsoft Is Selling More Than Cloud Credits
Microsoft Is Selling More Than Cloud Credits
The old startup-cloud pitch was simple: take credits, spin up infrastructure, and worry about enterprise architecture later. That bargain worked tolerably well for web apps, SaaS dashboards, and consumer experiments, where the first job was to prove product-market fit before the bills got serious. Biotech has never quite fit that model, because the “product” is often evidence, and evidence becomes less valuable when its lineage is murky.Microsoft’s new biotech framing recognizes that difference. The company is saying, in effect, that the startup stack now has to satisfy three audiences at once: scientists who need speed, engineers who need operational sanity, and external partners who need proof that sensitive data and regulated processes are under control. That is not merely a better Azure brochure. It is a bet that infrastructure choices will increasingly determine whether a biotech startup looks like a credible partner or an expensive science project.
There is a reason the language sounds like an operating-system metaphor. In modern biotech, the work is not just a sequence of experiments; it is a loop of hypothesis generation, model training, simulation, validation, documentation, and collaboration. If that loop lives across notebooks, ad hoc scripts, unmanaged clusters, and one-off file transfers, the company accumulates technical debt at the exact moment it is trying to accumulate scientific credibility.
Microsoft’s pitch is therefore aimed at a pain point founders already understand. The problem is not that biotech startups lack ambition. It is that a small team can be asked to behave like a pharmaceutical-grade computational organization before it has the headcount, procurement muscle, or process maturity to do so.
The Real Bottleneck Is No Longer Just Biology
Biotech has always been constrained by uncertainty, but the character of that uncertainty is changing. In an earlier era, the bottleneck was often access to wet-lab capability, assays, samples, or domain expertise. Those constraints have not disappeared, but they now sit beside a newer operational constraint: the ability to move between data, compute, models, and evidence without losing control of the process.That is why hyperscaler strategy matters in life sciences in a way it does not always matter elsewhere. A biotech startup running large simulations, training specialized models, analyzing clinical data, or collaborating with pharma is not merely renting servers. It is choosing the substrate on which its claims about reproducibility, security, and scalability will later be judged.
The Microsoft blog’s strongest point is that scientific ambition increasingly collides with infrastructure reality. Frontier workloads are bursty. A team may spend days iterating interactively, then suddenly need massive compute for simulation, molecular screening, model training, or evaluation runs. Hiring a full internal platform team too early can kill runway; postponing platform discipline too long can make the science hard to reproduce.
This is the paradox Microsoft is trying to exploit. Startups want the freedom of improvisation, but their customers and partners want the reassurance of discipline. The cloud provider that can make serious workflows feel less like bespoke infrastructure engineering has a stronger claim on the future biotech customer than one that merely offers the longest catalog of services.
AI-Native Biotech Is a Factory, Not a Chatbot
The least useful way to understand AI in biotech is as a chatbot bolted onto a research workflow. That framing understates both the opportunity and the risk. Scientific AI is not primarily about asking a model for an answer; it is about repeatedly constructing, testing, rejecting, and refining claims under conditions where errors can be expensive and misleading metrics can be worse than no metrics at all.Microsoft’s article leans into this reality by emphasizing iteration, fine-tuning, evaluation, lineage, and reproducibility. In biotech, generic models may help with literature review, coding, summarization, or workflow automation, but competitive advantage usually depends on proprietary data and specialized modeling choices. That means the startup’s system must support custom architectures, secure training and post-training, and controlled deployment paths rather than treating AI as a commodity API call.
The phrase AI-native gets abused in startup marketing, but here it has a concrete meaning. An AI-native biotech company is one whose experimental and computational loops are designed around models from the beginning. Its knowledge base, validation strategy, compute patterns, and governance processes are part of the same machinery.
That machinery also has to admit uncertainty. Model evaluation in science is not a leaderboard game. A metric can look impressive while masking dataset leakage, weak external validity, poor calibration, or a mismatch between academic benchmarks and biological usefulness. When investor pressure and publication incentives enter the room, the temptation to overstate model performance becomes structural.
This is where platform architecture becomes a form of scientific hygiene. A system that records how data moved, which model version produced which output, what evaluation protocol was used, and which assumptions were baked into a run is doing more than satisfying auditors. It is protecting the company from fooling itself.
Partnerability Is Becoming Architecture
The most commercially important idea in Microsoft’s post is partnerability. It is not a standard product-management term, but it should be. For many biotech startups, the path to scale runs through pharma, clinical research organizations, hospitals, data custodians, or regulated enterprise buyers. Those partners rarely want to ship their most sensitive data into a startup’s preferred environment just because the startup’s model performs well in a demo.That changes the architecture. A startup may need to deploy a containerized model inside a customer’s cloud tenant, keep the customer’s data in place, protect its own intellectual property, and still scale compute reliably. That is a harder problem than serving an API from a central account. It forces the startup to think about tenant boundaries, identity, secrets, logging, update mechanisms, and operational support long before a typical early-stage company wants to.
Microsoft’s advantage, if it can execute, is that Azure already lives inside many enterprise and regulated environments. That does not automatically make Azure the right answer for every biotech workload, but it does make the argument legible to buyers who care about data residency, identity integration, compliance posture, and procurement familiarity. The platform decision becomes a sales-enablement decision as much as a technical one.
The same logic applies to clinical and patient-derived data. Once protected health information enters the workflow, the cheerful language of AI experimentation gives way to access controls, audit trails, data minimization, and governance. Agentic systems raise the stakes further, because a tool that can plan, execute, and chain actions across data sources is also a tool that can create new failure modes if permissions and boundaries are sloppy.
This is the part of the AI boom that startup pitch decks often skate past. Autonomy is not free. The more agency a system has, the more important it becomes to define what it can touch, what it can change, what it can export, and who is accountable when it produces a plausible but wrong result.
Microsoft Discovery Turns the Pitch Into a Product Strategy
Microsoft Discovery is the most concrete product behind this biotech narrative. Introduced as an enterprise agentic platform for research and development, it combines specialized AI agents, a graph-based knowledge foundation, high-performance computing, and extensibility for an organization’s own tools and data. In Microsoft’s telling, it is not a general assistant but a system for running parts of the discovery loop itself.That distinction matters. A chat interface can help a scientist move faster, but it does not by itself create an R&D operating model. Discovery is designed around agents that can reason over knowledge, plan work, use tools, run analyses, and feed results back into an iterative process. The ambition is to move from “AI helps me write code” to “AI helps coordinate the research process.”
The opportunity is obvious. Scientific organizations are drowning in fragmented knowledge: papers, experimental records, internal reports, simulation outputs, assay results, datasets, and tacit process knowledge spread across teams. A graph-based knowledge engine, if implemented well, could give agents richer context than a pile of documents in a vector store. It could also make the reasoning trail more inspectable, which matters in environments where “the model said so” is not an acceptable explanation.
But the risk is equally obvious. The more Microsoft Discovery looks like an operating layer for R&D, the more customers will ask whether it can be trusted with the messy, consequential, regulated parts of science. Preview products can inspire imagination; production scientific systems have to survive procurement, validation, incident response, and skeptical domain experts.
For biotech startups, the practical question is not whether Microsoft Discovery is magical. It is whether the platform points toward a stack they should prepare for: agents as workflow participants, HPC as an on-demand substrate, proprietary data as a governed knowledge layer, and evaluation as a continuous process rather than a pre-demo ritual.
The Compliance Story Is Really a Speed Story
Microsoft’s emphasis on Azure’s GxP audit milestone is easy to dismiss as enterprise compliance theater. That would be a mistake. In life sciences, compliance work is not just a legal or quality function; it is a tax on speed, partnership, and credibility.GxP is shorthand for “good practice” quality guidelines and regulations across regulated life-sciences environments. For a biotech startup, the key point is not that a cloud provider can make the company compliant by association. It cannot. The point is that a platform with independently reviewed processes, documentation, controls, and regulated-workload posture can reduce friction when a startup must answer hard questions from pharma, CROs, clinical partners, investors, or quality teams.
That reduction in friction can be strategically meaningful. Early-stage companies do not have infinite cycles to spend on security questionnaires, infrastructure explanations, audit evidence, and bespoke deployment discussions. Every week spent reconstructing how a result was generated is a week not spent improving the underlying science.
This is why trust can become a speed advantage. A startup that can show clear lineage, controlled access, reproducible workflows, and a plausible regulated-workload path may move faster through partner diligence than one with a more impressive model but a weaker operational story. The market does not always reward the cleanest science first; it often rewards the company that can make its claims usable by institutions.
Microsoft’s framing is self-serving, of course. Azure is the product being sold. But the underlying claim is right: in biotech, the operational wrapper around AI increasingly determines whether AI can leave the demo environment and enter the enterprise.
The Startup Constraint Is the Whole Plot
The most credible part of Microsoft’s post is its repeated attention to early-stage constraints. Biotech startups do not usually fail because they forgot to buy enough cloud services. They fail because they run out of time, money, focus, or evidentiary momentum before the science becomes partnerable.A five-person computational team cannot afford to spend months reinventing secure infrastructure patterns. A founder cannot personally referee every data-access decision once collaborations multiply. A machine-learning scientist should not have to become a part-time cloud networking specialist simply to run distributed workloads at the frontier.
This is where the operating-model language earns its keep. The question is not whether Azure has a service for every problem. The question is whether a startup can assemble a coherent way of working from those services without drowning in configuration, integration, and governance overhead. Platforms win when they compress complexity without hiding the controls that regulated customers need.
There is a delicate balance here. Too little abstraction leaves scientists stuck in infrastructure glue. Too much abstraction creates black boxes that serious partners will not trust. The winning biotech platform has to make the common path easy while preserving enough transparency for audit, validation, and expert challenge.
Microsoft’s message to founders is that these choices should be made early. That is sound advice, but it cuts both ways. Choosing a hyperscaler deeply is a form of dependency, and dependency has costs. The same architecture that makes a startup easier to partner with in one enterprise ecosystem may make it harder to stay portable later.
The Cloud War Moves Up the Stack
For years, cloud competition was framed around compute, storage, databases, and AI accelerators. Those still matter, especially for expensive scientific workloads. But Microsoft’s biotech argument shows the battle moving up the stack into workflow, evidence, governance, and domain-specific operating models.This is not unique to biotech. Every industry is watching AI push cloud providers from infrastructure vendors into process vendors. The difference is that science exposes the stakes more clearly. A hallucinated slide summary is annoying; a poorly governed model-driven discovery pipeline can distort research priorities, waste lab resources, or produce claims that crumble under external scrutiny.
Microsoft is not alone in seeing this opportunity. Other hyperscalers and specialist platforms are also chasing life sciences with managed data environments, research clouds, AI tooling, and compliance-oriented services. The market will not be won by press releases about agents. It will be won by the tedious details: identity integration, container deployment, data controls, cost predictability, observability, validation support, and the ability to meet scientists where they actually work.
Still, Microsoft has a distinctive strategic opening. Its enterprise footprint gives it a familiar route into pharma and regulated organizations. Its AI partnership and Azure AI stack give it model-building credibility. Its productivity software gives it a natural surface area for knowledge work. Microsoft Discovery attempts to connect those assets into something more vertical than generic cloud infrastructure.
That is the bigger story behind the startup blog. Microsoft is not merely asking biotech founders to apply for credits. It is trying to define what a credible AI-first biotech company should look like — and then position Azure as the place where that company is easiest to build.
Agentic Science Needs Guardrails Before It Needs Hype
The word agentic is doing a lot of work in 2026. In some contexts, it means little more than a chatbot with tool access. In scientific R&D, the term should be held to a higher standard, because a system that proposes hypotheses, launches computations, rewrites code, or generates documentation is participating in the production of evidence.Microsoft’s post wisely avoids telling founders simply to “adopt agents.” The better advice is to architect for agentic workflows deliberately. That means treating transparency, traceability, permissions, containment, and human oversight as design requirements rather than compliance afterthoughts.
A useful scientific agent should be able to show its work. It should expose which data it used, which tools it called, which assumptions it made, and where human approval entered the loop. It should be constrained by role and context, not handed broad access because the demo looked impressive.
This will matter even more as agents move from code generation and workflow automation into semi-autonomous execution. In a biotech setting, an agent might generate analysis scripts, select datasets, trigger simulations, summarize literature, prepare reports, or suggest next experiments. Each of those tasks can save time. Each can also introduce errors that look polished enough to pass casual review.
The lesson for founders is straightforward: autonomy without governance is not a platform advantage. It is a liability waiting for diligence.
The Evidence Package Becomes the Product
One of the most useful concepts in Microsoft’s post is the idea that startups should curate an “evidence package.” That phrase deserves more attention than it gets. In biotech, the asset being sold or partnered is not only a molecule, model, assay, or platform. It is the accumulated case that the work is real, reproducible, and worth trusting.An evidence package includes results, but it also includes context. What data was used? What transformations were applied? What model version generated the output? What evaluation criteria were chosen? What failed? What was repeated? What can an external party inspect without compromising intellectual property?
This is where many AI-first companies outside life sciences have been allowed to be vague. Biotech does not offer the same luxury. If a startup wants to influence a development program, support a clinical path, or persuade a pharma partner to run deeper validation, it needs more than a confident dashboard. It needs a chain of custody for scientific claims.
The irony is that this kind of discipline can feel slow at the beginning. Founders want velocity, and process feels like drag. But poor provenance creates a balloon payment later, when a partner asks for evidence and the team has to reconstruct the past from notebooks, Slack threads, storage buckets, and memory.
Microsoft’s platform argument is strongest when it frames reproducibility not as bureaucracy but as acceleration. If evidence is the product, then the system that produces, records, and packages evidence is core product infrastructure.
What WindowsForum Readers Should Notice About This Cloud Pitch
For Windows enthusiasts and IT pros, the biotech angle may seem distant from the daily concerns of endpoint management, Microsoft 365, identity, and Azure administration. It is not. The same enterprise patterns that reshaped ordinary IT are now moving into scientific computing: centralized identity, governed data access, audit trails, containerized deployment, policy enforcement, and managed infrastructure that still has to satisfy demanding users.The scientific workstation is becoming part of a larger governed fabric. Researchers may still live in Python, notebooks, specialized tools, and lab systems, but the environment around them increasingly resembles enterprise IT. That creates opportunities for sysadmins and cloud architects who understand both operational discipline and user friction.
It also creates a warning. If IT turns agentic science into a locked-down maze, researchers will route around it. If researchers ignore governance, partners and regulators will eventually force a reckoning. The winning organizations will be the ones that make the secure path the usable path.
Microsoft’s biotech message is therefore part of a broader Microsoft story. The company wants Azure, identity, security, AI tooling, and domain-specific platforms to become the default control plane for high-value work. In biotech, that work happens to be drug discovery, clinical evidence, and scientific R&D. In other sectors, the same pattern will show up as engineering, finance, legal operations, manufacturing, and design.
Microsoft’s Biotech Bet Comes With Strings Attached
There is a real strategic trade-off for founders. Building deeply on Azure can make enterprise conversations easier, especially when prospective partners already trust Microsoft’s identity, security, and compliance ecosystem. It can also concentrate architectural assumptions around one vendor’s services, pricing, roadmap, and abstractions.That does not mean startups should avoid hyperscaler-native design. In many cases, avoiding it is the riskier move. A fragile platform assembled from unmanaged scripts and heroic engineering effort is not more independent in any meaningful sense. It is merely dependent on people and practices that may not scale.
But founders should be clear-eyed. The right platform strategy preserves leverage where it matters. Containerized workloads, well-defined data boundaries, exportable evidence, documented evaluation pipelines, and clean interfaces can help a startup benefit from Azure without turning every future partnership into a rewrite.
This is especially important in pharma, where the customer’s cloud environment may not match the startup’s preferences. A credible “run where the data lives” story should not be an afterthought. It should be part of the product architecture from the beginning, because enterprise adoption often depends on deployment patterns as much as model performance.
Microsoft is correct that platform is strategy. The corollary is that platform strategy should be negotiated, not sleepwalked into.
The Azure Biotech Playbook Is Really an Operating Discipline
Microsoft’s post is nominally aimed at biotech founders, but its most concrete lessons are useful well beyond the startup audience. They describe the shape of a modern scientific computing organization: elastic compute, secure collaboration, repeatable pipelines, disciplined evaluation, and agentic workflows constrained by governance.The core takeaways are practical rather than mystical:
- Startups should design for bursty scientific workloads, because the shift from interactive exploration to large-scale compute is now a normal part of AI-enabled biotech.
- Proprietary data and specialized models remain central to scientific advantage, so platforms must support fine-tuning, validation, lineage, and secure deployment rather than only generic model access.
- Pharma-readiness increasingly depends on architecture, including tenant-aware deployment patterns that keep sensitive customer data under customer control.
- Agentic workflows should be introduced with explicit permissions, traceability, containment, and human oversight, especially when agents can execute code or act across shared datasets.
- Reproducibility and auditability should be treated as product features, because partners and regulators will judge the evidence trail as well as the headline result.
- Cloud compliance milestones do not make a startup compliant by default, but they can reduce diligence friction when the startup’s own controls are well designed.
Biotech’s AI future will not be decided by the most theatrical agent demo or the most extravagant claim about replacing scientists. It will be decided by which companies can turn computation into credible evidence again and again, under the scrutiny of partners who own the data, regulators who demand traceability, and scientists who know how easily biology humiliates overconfident software. Microsoft’s wager is that Azure can become the operating system for that discipline; founders should treat the pitch seriously, but not passively, because the platform choices they make now may define not only how fast they move, but whether anyone believes the results when they arrive.
Source: Microsoft Biotech’s AI upgrade: An operating system built for science