INBRAIN Neuroelectronics and Microsoft have announced a strategic collaboration to fuse INBRAIN’s graphene-based brain-computer interface (BCI) therapeutics platform with Microsoft’s Azure cloud, large language models (LLMs) and emerging agentic AI tooling — a move the partners say could accelerate real-time, closed-loop, adaptive therapies for neurological disorders such as Parkinson’s disease, epilepsy, and memory or psychiatric conditions.
INBRAIN Neuroelectronics, a Barcelona‑based spin‑out focused on ultrathin graphene neural interfaces and AI‑driven neuromodulation, has been building a clinical pathway for what it calls “BCI‑Tx” — brain‑computer interface therapeutics — and recently completed a significant Series B financing round while reporting first‑in‑human activity in clinical trials. The company also highlights that its platform has received an FDA Breakthrough Device designation for Parkinson’s disease-related applications. Microsoft’s involvement centers on providing Azure’s cloud compute, data architecture, and AI tooling — including LLMs and agentic AI frameworks — to enable continuous learning, time‑series analysis of neural signals, and the software backbone for real‑time adaptive closed‑loop therapy. Microsoft executives framed the work as part of a broader push to bring agentic AI into regulated and high‑stakes industries while emphasizing responsible AI principles for healthcare. This collaboration is notable because it pairs a materials‑driven BCI hardware specialist with one of the largest global cloud and AI providers. If successful, it could shift current neuromodulation and neurostimulation models from periodic clinical tuning to continuous, software‑driven personalization that adapts therapy in milliseconds to minutes based on patient state.
If the collaboration manages the engineering and ethical challenges effectively, it could reshape neuromodulation from discrete interventions to continuous, personalized therapies that behave more like software‑defined organs than fixed medical devices. Until then, cautious optimism is the right stance: the promise is real, the technical groundwork is under way, and the next 12–36 months of clinical data and architectural detail will be crucial in making that promise concrete.
Source: Windows Report INBRAIN & Microsoft Partner to Develop AI-Powered Brain Interface Tech (BCI)
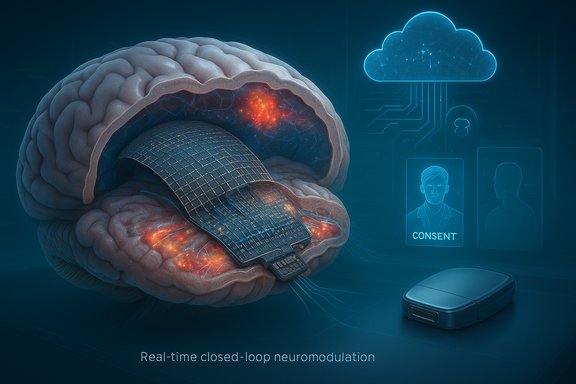 Background / Overview
Background / Overview
INBRAIN Neuroelectronics, a Barcelona‑based spin‑out focused on ultrathin graphene neural interfaces and AI‑driven neuromodulation, has been building a clinical pathway for what it calls “BCI‑Tx” — brain‑computer interface therapeutics — and recently completed a significant Series B financing round while reporting first‑in‑human activity in clinical trials. The company also highlights that its platform has received an FDA Breakthrough Device designation for Parkinson’s disease-related applications. Microsoft’s involvement centers on providing Azure’s cloud compute, data architecture, and AI tooling — including LLMs and agentic AI frameworks — to enable continuous learning, time‑series analysis of neural signals, and the software backbone for real‑time adaptive closed‑loop therapy. Microsoft executives framed the work as part of a broader push to bring agentic AI into regulated and high‑stakes industries while emphasizing responsible AI principles for healthcare. This collaboration is notable because it pairs a materials‑driven BCI hardware specialist with one of the largest global cloud and AI providers. If successful, it could shift current neuromodulation and neurostimulation models from periodic clinical tuning to continuous, software‑driven personalization that adapts therapy in milliseconds to minutes based on patient state.Why graphene matters: the technology foundation
Graphene as a neural interface material
Graphene — a single atom‑thick sheet of carbon atoms — is prized for exceptional electrical conductivity, mechanical strength, flexibility, and a very high surface‑to‑volume ratio. These properties make it attractive for neural interfaces that must be conformal, minimally invasive, and capable of high‑density recording and stimulation. Academic reviews and preclinical studies have explored graphene’s mechanical, thermal, and electrical properties and its potential biocompatibility when used in neuro‑electronic implants. INBRAIN’s publicly stated device features include ultrathin, high‑density graphene electrode arrays and a compact neural processor designed for implantable operation — the company claims micro‑scale elements with 10–100× higher spatial resolution than many existing neuromodulation electrodes. Those device characteristics are central to the company’s pitch for enabling fine‑grained decoding of pathological neural patterns.What graphene brings to closed‑loop therapy
- Higher spatial resolution — enables targeting small nuclei and micro‑circuits in deep brain structures.
- Lower impedance / higher charge injection capacity — can reduce power demands and enable smaller, denser arrays.
- Flexibility and thinness — may reduce foreign‑body response and improve chronic signal stability.
What Microsoft brings: cloud, LLMs, and agentic AI
Azure and healthcare AI building blocks
Microsoft will contribute Azure cloud infrastructure, data governance tools, and its LLM ecosystem (Azure OpenAI & related services) to host the continuous learning and analytics pipeline that INBRAIN envisions. The collaboration announcement specifically references time‑series LLM capability and agentic AI (AI agents that operate with a degree of autonomy to monitor, reason about, and act on streaming data) as core enablers of adaptive neuromodulation. Microsoft has already been positioning agentic AI and Copilot‑style agent frameworks for regulated domains — including healthcare agent services and Copilot Studio components designed to embed safety and compliance controls — which suggests the company will reuse, adapt, and extend these governance primitives when working with INBRAIN.Why LLMs and time‑series models matter for neural data
Neural recordings are fundamentally time‑series data: high‑frequency, multi‑channel waveforms containing patterns across milliseconds to minutes. Recent research shows that LLMs and large sequential models can be repurposed for time‑series forecasting, anomaly detection and multi‑modal reasoning when properly adapted — a growing area of research across industry and academia. Translating LLM reasoning to neural data could allow models to detect transient biomarkers, predict seizure onset, or infer behavioral states from complex spatiotemporal patterns. However, important caveats apply: LLM‑based approaches to time series are an active research area, and their robustness in safety‑critical, biological closed‑loop control remains unproven at scale. The adaptation requires rigorous domain alignment, low‑latency inference, interpretable outputs, and continuous validation against clinical outcomes.The clinical promise: what closed‑loop AI‑enabled BCI could do
Potential therapeutic applications
- Parkinson’s disease — continuously detect pathological oscillations and apply graded stimulation to reduce motor symptoms while minimizing stimulation‑related side effects.
- Epilepsy — detect pre‑ictal signatures and deliver precisely timed interventions to abort or blunt seizures.
- Mood and psychiatric disorders — identify state biomarkers and deliver targeted modulation to network nodes implicated in depression, OCD, or PTSD.
- Memory disorders — support closed‑loop cues or stimulation that enhance encoding or retrieval circuits.
How closed‑loop could change outcomes
Closed‑loop systems promise three core improvements over open‑loop stimulation:- Efficacy improvement — delivering therapy only when biomarkers indicate need.
- Side‑effect reduction — limiting off‑target stimulation or overtreatment.
- Energy efficiency — lowering battery drain and enabling smaller implants.
Technical and clinical challenges — a reality check
Latency, reliability, and on‑device compute
Real‑time closed‑loop neuromodulation requires extremely low‑latency detection and action pathways. Cloud connectivity adds complexity: while Azure can host heavy training and analytics workloads, latency and reliability constraints generally push decision‑critical inference and fail‑safe controls to an on‑device or edge layer. Achieving a safe split between on‑device real‑time controllers and cloud‑based continuous learning systems will be central to viability.Model drift and continuous learning
Neural signals change over time due to plasticity, electrode encapsulation, medication changes, and disease progression. Continuous learning systems must detect and manage concept drift — updating models without introducing destabilizing behavior. Guardrails, human‑in‑the‑loop oversight, and periodic clinical revalidation will be required. Academic work on LLMs for time‑series highlights both promise and the need for domain‑specific adaptation strategies.Interpretability and regulatory acceptance
Regulators and clinicians demand interpretable decisions for therapies that directly interact with the nervous system. Black‑box LLM outputs will be scrutinized; explainability, audit trails, and reproducible decision logs will be necessary for approval and adoption. The FDA Breakthrough Device designation speeds interaction with regulators but does not reduce the need for robust evidence of safety and effectiveness.Safety and adversarial risks
Any networked implant that incorporates remote, adaptive intelligence introduces novel safety surfaces: cyber‑security attacks against the cloud or device, adversarial inputs that could mislead models, and software bugs that alter stimulation incorrectly. These require multi‑layered defenses: hardware isolation, encrypted telemetry, tamper‑resistant firmware, and independent watchdog controllers that can halt therapy if anomalous behavior is detected.Biocompatibility and long‑term device reliability
Graphene’s material promise is real — but long‑term biocompatibility, chronic inflammatory response and mechanical durability in a live human brain remain active areas of clinical evaluation. Early human trials are encouraging on feasibility, yet chronic safety, signal stability over years, and explant scenarios must be proven in larger cohorts.Regulation, data governance and ethics
Regulatory pathways and evidence expectations
- The FDA’s Breakthrough Device program offers priority engagement and advice, but clearance or approval will still require rigorous clinical evidence demonstrating safety and clinical benefit.
- For any AI‑enabled therapy with autonomous elements, regulators will expect lifecycle management plans, risk mitigation strategies for model updates, and documented validation of the entire hardware‑software chain.
Data privacy and cross‑border data flows
Neural data is some of the most sensitive personal data imaginable. Patients and regulators will insist on stringent privacy protections: de‑identification, differential privacy where feasible, strict access controls, and clear consent frameworks for data used to train cloud models. Microsoft’s enterprise healthcare tooling includes compliance scaffolding, but deployment across jurisdictions will require localized controls and legal safeguards.Ethical guardrails for agentic AI
Agentic AI implies autonomous agents making or recommending actions. For brain‑connected systems, ethical concerns intensify: patient agency, informed consent for autonomous behaviors, the right to an “off” state, and transparency about when AI acts without real‑time clinician input. Developers will need to operationalize consent models, incorporate human overrides, and prioritize modes that maintain patient control.Business and strategic implications
Why Microsoft is investing
For Microsoft, the partnership advances multiple strategic goals: expanding Azure’s high‑value healthcare footprint, demonstrating agentic AI in regulated settings, and building verticalized solutions that lock in long‑term cloud revenue and services. Healthcare partnerships also provide narratives of socially beneficial AI deployment — a reputational asset amid scrutiny over AI risks.What INBRAIN gains
INBRAIN gains access to scalable compute, compliance tooling, and a broad enterprise ecosystem — accelerating the path from bench to clinic and enabling heavy‑compute model training that would be otherwise costly or slow for a small med‑tech startup. The partnership also signals investor confidence and could smooth procurement conversations with hospitals and health systems that already use Microsoft products.Competitive landscape
Other players in neurotechnology — major med‑tech firms, academic spinouts, and defense‑funded initiatives — are also pursuing closed‑loop neuromodulation and BCI therapeutics. Microsoft’s cloud muscle could be a differentiator for INBRAIN, but incumbents with large regulatory and clinical trial experience remain formidable. Partnerships with large pharma (e.g., INBRAIN’s reported collaborations) and imaging/clinical partners will be crucial to scale clinical validation.Roadmap to deployment: practical steps and timeline considerations
Below is a condensed pathway from announcement to potential patient impact, presented as a practical checklist that highlights dependencies where Microsoft and INBRAIN will need to demonstrate capability and trustworthiness.- Preclinical & device validation — chronic implant safety, hermetic sealing, signal stability testing.
- Pilot human studies — feasibility, acute safety, short‑term efficacy signals (ongoing for INBRAIN).
- Regulatory dialogue — staged evidence packages, risk mitigation plans for adaptive algorithms (Breakthrough Device designation supports this step).
- Architecture design — split computation: deterministic on‑device safety controller + cloud‑based continuous learning pipeline.
- Clinical trials (pivotal) — randomized, controlled studies measuring clinically meaningful outcomes and safety across diverse populations.
- Post‑market monitoring — long‑term surveillance, model update governance, and cyber‑resilience testing.
- Healthcare integration — EHR interoperability, clinician workflows, reimbursement coding and payer evidence generation.
Strengths of the partnership
- Complementary expertise — INBRAIN’s graphene BCI hardware + Microsoft’s cloud/AI stack is a strong technical fit for an ambitious closed‑loop system.
- Regulatory engagement — the Breakthrough Device designation gives the teams prioritized access to FDA guidance, accelerating iterative testing and submission cycles.
- Scalable compute and governance — Azure provides global compliance tools, identity management, and trusted enterprise relationships that med‑tech companies need to reach large hospital systems.
- Research synergy — new approaches to time‑series with LLMs could offer richer, multi‑scale pattern recognition than classical signal‑processing alone.
Key risks and unresolved questions
- Autonomy versus safety — how much autonomy will agentic AI be allowed in closed‑loop control, and what failsafe architectures will be mandated? These questions have clinical and ethical weight that are not solved by technology alone.
- Latency and on‑device needs — heavy reliance on cloud models may hinder real‑time responsiveness unless critical control loops remain on‑device.
- Model robustness — neural signals are noisy and non‑stationary; LLM/time‑series methods must be validated under many physiological conditions to avoid harm.
- Data governance and privacy — neural telemetry requires stronger-than‑usual protections; cross‑border data flow for model training will face regulatory friction.
- Hacking and adversarial threats — the consequences of a malicious actuator command are severe; security design must be proven as part of clinical evidence packages.
What to watch next
- Progress in INBRAIN’s ongoing clinical trials and any published interim or pivotal results that quantify efficacy, safety, and signal stability.
- Technical papers or demos showing how LLMs or agentic frameworks can operate on neural time‑series data with the latency and interpretability required for closed‑loop control.
- Regulatory filings and FDA interactions that clarify how autonomous AI components will be handled in device submissions.
- Security audits or third‑party evaluations addressing cyber‑risk management for networked implants.
Conclusion
The Microsoft–INBRAIN collaboration is a high‑profile step toward integrating advanced materials, neural engineering and cloud‑scale AI into a single therapeutic stack for brain disorders. The technical rationale is sound: graphene interfaces can deliver higher resolution signals and Microsoft’s Azure and agentic AI tooling can provide the compute and continuous learning infrastructure necessary for adaptive therapy. But the pathway from press release to durable, safe, scalable clinical outcomes is long and complex. Key technical hurdles — low‑latency control, model robustness, long‑term biocompatibility — and non‑technical barriers — regulatory acceptance, privacy, and cyber‑security — will determine whether this partnership becomes a durable platform or a promising experiment.If the collaboration manages the engineering and ethical challenges effectively, it could reshape neuromodulation from discrete interventions to continuous, personalized therapies that behave more like software‑defined organs than fixed medical devices. Until then, cautious optimism is the right stance: the promise is real, the technical groundwork is under way, and the next 12–36 months of clinical data and architectural detail will be crucial in making that promise concrete.
Source: Windows Report INBRAIN & Microsoft Partner to Develop AI-Powered Brain Interface Tech (BCI)